 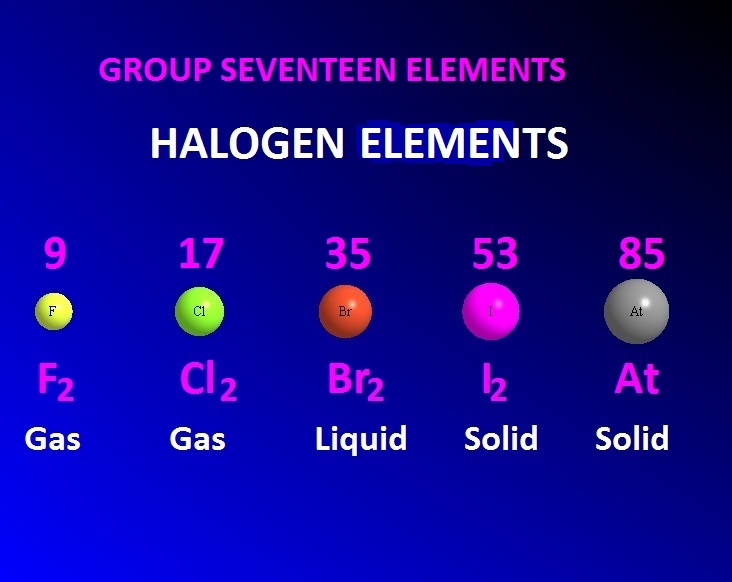
Iodine will sublime directly from a solid into a gas.Chlorine gas killed thousands of soldiers during world war I in 1915, when used as a chemical weapon.It not only damages the body on the outside but it can be absorbed through the skin, penetrating deep into tissues causing severe systemic injury Although hydrofluoric acid, HF, is a weak acid, it is one of the most dangerous acids to be around.All isotopes are radioactive and short-lived. A sample of pure astatine has never been assembled. Astatine is the rarest element that occurs naturally in the earth’s crust.Iodate ions play a key role in many “clock” reactions used for chemical demonstrations.Bromide and bromate ions are key ions in a famous and beautiful oscillating reaction known as the Belousov–Zhabotinsky reaction.Students learn that silver (I) chloride is insoluble, but most people don’t know that copper (I) chloride is also insoluble in water.Many rare-earth fluorides are insoluble in water.The halogens will rip an electron from another atom, in order to achieve a very stable state of 8 electrons in their outer shell. Halogens are so reactive, because they have 7 valence electrons, and are very close to having a complete shell of 8 electrons. Halogens also form many different interhalogen compounds, for example IF 7, iodine (VII) fluoride. Fluorine commonly occurs in minerals like fluorite, CaF 2. They also form anions like the hypochlorite, chlorate, perchlorate, and bromate ions that are very strong oxidizing agents. The halogens react violently with alkali metals, alkaline earth metals, and aluminum, especially when there is a large surface area of metal. In this article, we focus just on the first four halogens. The halogen family includes fluorine, chlorine, bromine, iodine, astatine, and tennessine, element 117 – the most recent element to be discovered. The halogens all have an interesting color, which can change depending on the medium or what it is dissolved in, see a detailed explanation here. 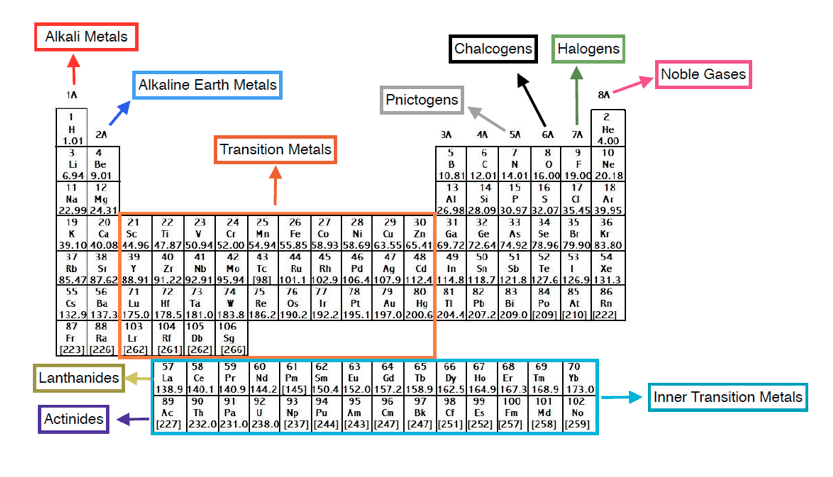
The first ionization energy of fluorine is so high, that it does not exhibit any positive oxidation states. The ionization energy also decreases as you move down, making it easier to form higher oxidation states. In fact, fluorine has the highest electronegativity of any element. All of the halogens have a high electronegativity, but it decreases as you move down the column. It is the only group on the periodic table that has elements in the solid, liquid and gas phase at room temperature. So fluorine is a gas, and iodine is a solid. The boiling points also increase due to increased Van Der Waals forces. The fluorine-to-fluorine bond is the weakest 
In the elemental state, they form diatomic molecules, joined by nonpolar covalent bonds. Moving down the column, halogens increase in atomic radius as they add additional electron shells. Because of their reactivity, there are almost never found in nature in their elemental form. These unique properties are because of the 7 electrons in their valence shell. Breathing in vapors of any of the halogens is very dangerous, and they have distinctive, unpleasant odors (although the author finds a faint smell of bromine oddly interesting).įluorine is the most reactive of all elements, and can only be stored in metals that form passivation layers of the fluoride salt. The halogens are highly reactive and highly toxic. The Greek word “hal”, meaning salt, also appears in the name of the mineral halite, aka sodium chloride. In 1826, Swedish chemist Jons Berzelius coined the term halogen for the entire group of elements. In 1811 John Schweigger wanted to name the element chlorine “halogen”, but he failed. They are salt-producing, because when they react with metals (often violently), they produce ionic compounds known as salts. They are non-metals that consist of diatomic molecules. Fluorine and chlorine are the “poster children” of the halogens. The halogens, aka halogen family, are a group of reactive elements in group 17 of the periodic table, to the right of the chalcogens, and to the left of the noble gases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
